FDA Advisory Skepticism: mpathic Addresses Psychedelic Clinical Trials Concerns
In recent deliberations, the FDA’s Psychopharmacologic Drugs Advisory Committee (PDAC), decided against the approval of MDMA-assisted therapy (MDMA-AT) for the treatment of post-traumatic stress disorder (PTSD) by study sponsor, Lykos Therapeutics. Although the vote is nonbinding, it significantly influences the FDA’s decision-making process. The advisory committee’s stance reflects substantial reservations concerning the safety and the methodology underpinning the clinical trials investigating the therapeutic benefits of MDMA-assisted therapy. Such recommendations, particularly coming from this influential panel, play a crucial role as the FDA approaches its final decision, expected by August 11, 2024.
The advisory committee found that the available data were insufficient to demonstrate the effectiveness of MDMA-assisted therapy (MDMA-AT) for PTSD in a 9-2 vote. Furthermore, in an even more decisive 10-1 vote, they concluded that the benefits of MDMA-AT do not outweigh the potential risks associated with its use.
In the chart below, explore the detailed concerns flagged by the FDA committee and examine how mpathic, a leader in AI-driven conversation analytics, can address these challenges to enhance both the efficacy and safety of clinical trials. This analysis will cover a range including standardizing methodologies to address biases and prevent misconduct, enhancing the safety of participants, and evaluating the pivotal role of psychological support/therapy.
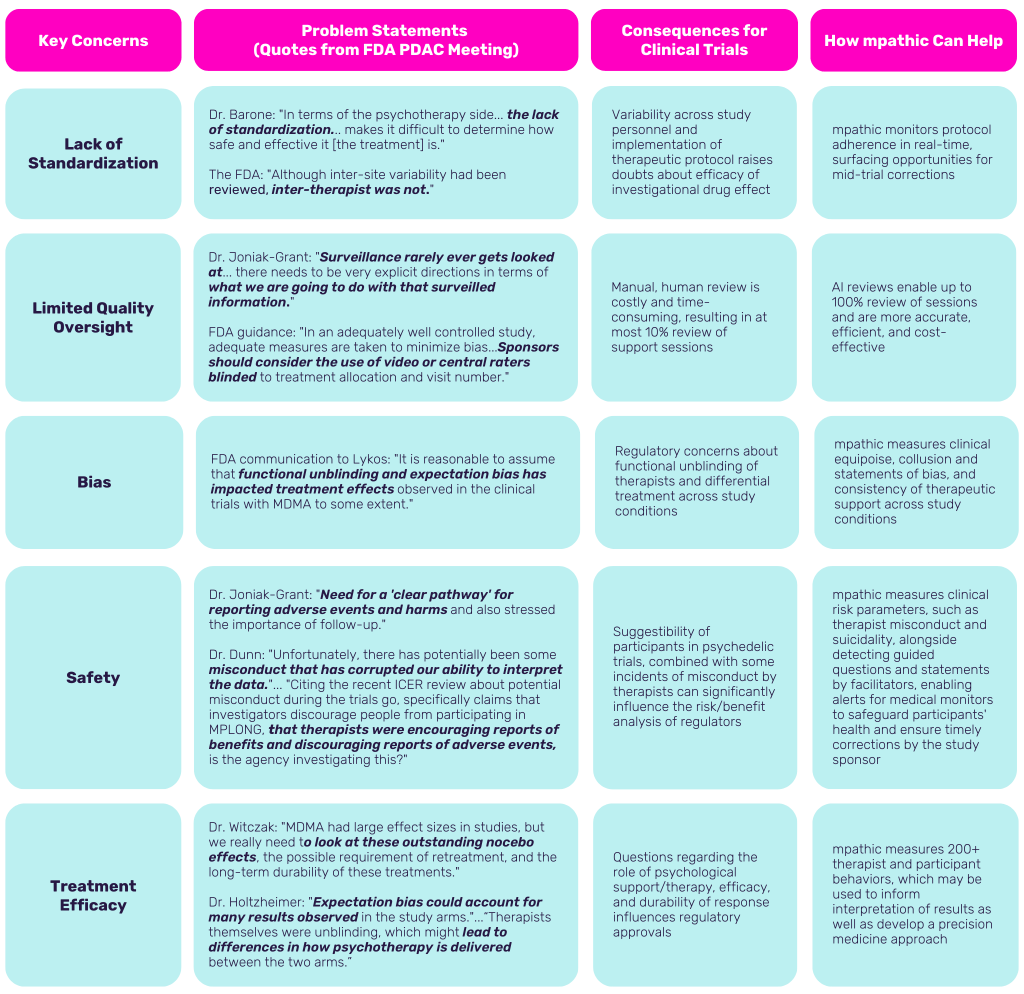
These deliberations highlight the pressing need for enhanced oversight and consistency in clinical trials. The committee’s reservations emphasize the challenges that an innovative approach like mpathic’s AI-enabled conversation analytics, through its mTrial platform, is particularly well-suited to address.
mpathic’s mTrial enhances the scale and consistency of traditional trial oversight by reviewing 100% of psychological support sessions using AI fidelity monitoring, achieving a 7x accuracy rate over human raters. This level of analysis provides robust assurance of the quality of data and the adherence to protocols that the advisory committee is rightfully expecting. Moreover, mTrial capabilities include providing objective reports for regulatory review while maintaining the required independence and blinding for sponsors, ensuring that the integrity of data is preserved from any biases.
mpathic’s response to the need for quality and standardization is paramount in mitigating the risks of unblinding and expectancy effects, which were some key concerns from the advisory committee. By providing a real-time response system, mTrial promotes consistency in therapeutic delivery throughout the trial, mitigating variability and contributing to the overall success rates of these groundbreaking treatments.
We at mpathic remain hopeful and committed to serving as a strategic partner within the psychedelic research community. As the market leader in technology-assisted oversight, serving the majority of psychedelic sponsors, we are dedicated to assisting trial sponsors in not just meeting but exceeding FDA’s recommendations. Our involvement ensures that future trials are conducted with the utmost safety and consistency, providing much-needed support for those suffering from mental health problems and other CNS disorders. The future of psychedelic research is, without a doubt, more robust with mpathic as an integral ally. With tools like mTrial, the path to regulatory approval becomes clearer and more navigable, paving the way for more effective treatments and improved outcomes for patients worldwide.
Interested in learning more about the cutting-edge methods and standards discussed? Discover how mpathic’s platform can transform how psychedelic clinical trials are monitored. Click the button below to explore our solutions and book a demo today.
